Advantages
- Highly efficient
- Flexible installation
- Wide range of applications
- Minimal emission of noise and pollution
- Immense potential for further technological development
Unlike traditional generation techniques, fuel cells turn fuel into energy without having to generate heat first. Check out the characteristics, types and possible applications of this high-end technology!
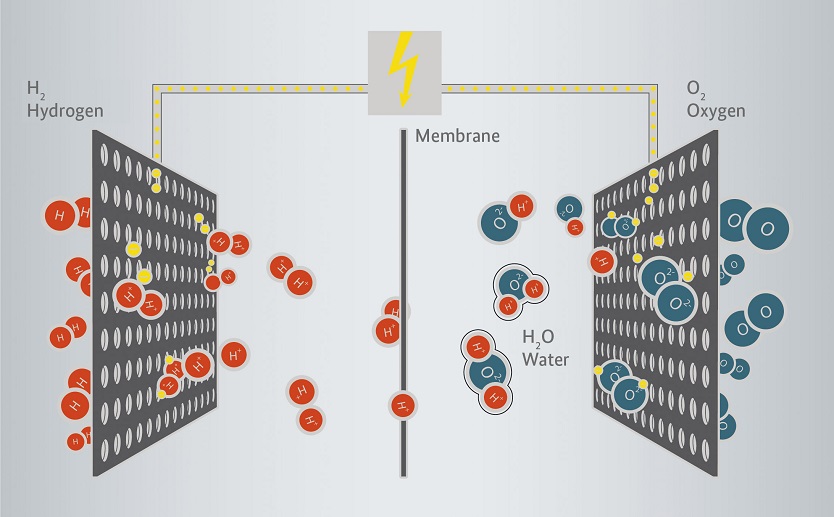
Operating principle of a fuel cell
© Viessmann
Fuel cells offer a very promising avenue for sustainable generation of electricity and heat. The technology is based on the electro-chemical reaction that occurs when hydrogen ions meet oxygen ions. Heat, water vapour and direct current are produced when this happens – without any noise or waste gases that have an impact on climate.
Fuel cell technology is CO2-neutral if the hydrogen is produced with renewable electricity via electrolysis. It has proven itself effective in practice, can be systematically regulated and is highly effective.
In residential buildings, fuel cells act as small-scale power plants to supply electricity and heat. Emission-free hydrogen-powered buses and trains are already being used for passenger transport in Germany. These virtually generate their own energy directly on-board.
Market development in Germany is making good progress. While just under 2,000 stationary fuel cells were produced in 2016, this is expected to rise to more than 90,000 large plants and 20,000 smaller systems by 2022 (see IWR Online, in German). German research institutes and companies have grappled with this technology for decades and have acquired extensive knowledge and expertise.
Fuel cell technology has the potential to revolutionise the energy world.
In a chemical process referred to as “cold burning”, a fuel cell transforms oxygen and hydrogen or other fuels (i.e. methanol, diesel, etc.) into energy, heat and water. Unlike most conventional technologies, the energy generation works directly, without the production of steam and the use of turbines. That is why the application of the fuel cell technology could significantly improve energy efficiency.
In fuel cells, heat and electricity are generated from an electrochemical reaction. The core components of a cell are an anode, cathode and electrolyte membrane. Hydrogenous gas is delivered to the anode, while oxygen is delivered to the cathode. To prevent a direct oxyhydrogen reaction, the two gases are separated from each other by a membrane which is impermeable to gas. Free electrons and positively charged hydrogen ions are formed at the anode and diffused through the electrolyte membrane. Here they meet the negatively charged oxygen ions which are formed at the cathode. Both types of ions react chemically in an exothermic reaction to water while heat is released. The free negatively charged electrons formed on the anode side are discharged along an external conductor and used as active current in the form of direct current.
Fuel cells with membranes function at temperatures of 70 to 90° C. A polymerelectrolyte-membrane-fuel cell or low-temperature fuel cell is known as a “proton exchange membrane fuel cell“. In future, this type of fuel cell will be increasingly used to power vehicles or as a highly efficient source of power and heat in individual buildings.
High-temperature fuel cells, on the other hand, function at temperatures of 900 to 1,000° C and comprise solid oxide electrolytes to separate hydrogen and oxygen in the cell. At this temperature range, fuel cells can be operated as highly efficient heat and power stations for generating heat for local heating networks. High-temperature fuel cells are also known as“solid oxide fuel cells“.
Fuel cells can be applied in a wide range of areas and cases: Car manufacturers use fuel cells to power their vehicles. This technology could compete with both, traditional combustion engines and electric vehicles. Many automotive producers have already initiated programs for realising fuel cell engine technologies.
Stationary appliances, such as heating systems for homes and other buildings, are another promising area for fuel cell rollout.
In addition, fuel cells could compete with rechargeable batteries when it comes to powering small mobile devices such as smart phones, tablets or laptop computers.
energy solutions - made in Germany
Technology brochure
Opens PDF "energy solutions - made in Germany" in a new window.